The transportation equation

Xuesong Zhou, an associate professor of civil and environmental engineering in the Ira A. Fulton Schools of Engineering, has spent a majority of his career working to create a universal mapping system for transportation researchers to use to look at the multiple levels of interaction that happen between cars, bikes and pedestrians in complex networks. Image courtesy Xuesong Zhou/ASU
Xuesong “Simon” Zhou says there are several variables that make up his vision for an ideal transportation system of the future.
First is creating a carbon neutral environment by 2050. The second involves improving access to transportation for disabled, low-income and other minority groups to create equity with the driving population.
These are not easy feats on their own and for these changes to be effective, they must happen at the same time. This presents even more of a challenge for transportation researchers like Zhou.
Before about 1940, walking, bicycling and public transit were recognized as important travel modes, but for most of the last century, transport planning has been automobile oriented. As a result, most communities now have well-developed road systems that allow motorists to drive to most destinations safely and with relative convenience; at worst they may be delayed by peak period congestion or pay tolls and parking fees at some destinations. However, such planning ignored the needs of people who use non-automobile travel modes.
Zhou, an associate professor focusing on multimodal transportation research in the School of Sustainable Engineering and the Built Environment, one of the seven Ira A. Fulton Schools of Engineering at Arizona State University, says that is where his work comes into play.
“We need to have a seamless connection between design planning and execution,” Zhou says. “The two steps are reliant on one another, so if we plan for an effective implementation, we plan for a better design and vice versa.”
Cars, buses, bikes and pedestrians
Zhou’s multimodal transportation research investigates how various transportation options play an integral role in meeting the mobility needs of the populations that rely on them, how each transit option impacts the usage of other modes of transportation as well as the infrastructure needed to support the multiple transportation options.
Urban intersections in the United States have many of the same components, such as a number of lanes utilized by vehicles or buses, other lanes for bikes and sidewalks for pedestrians. Each element represents a critical component of a multimodal network that needs to be considered when creating models and planning out cities.
In recent years, Zhou has worked with numerous transportation researchers and practitioners to find ways to streamline research in the field of multimodal transportation modeling. In most instances, researchers create their own data sets and unique maps for the regions where they conducted their research. As a result, when researchers from other areas want to replicate the study design in their region, they need to start from scratch.
“There was no universal system for building models and the networks upon which they are built,” Zhou says. “We created a way to convert General Transit Feed Specification, or GTFS, and OpenStreetMaps, which are industry standards, into the General Modeling Network Specification, or GMNS, a common human and machine readable format that can be shared between transportation researchers.”
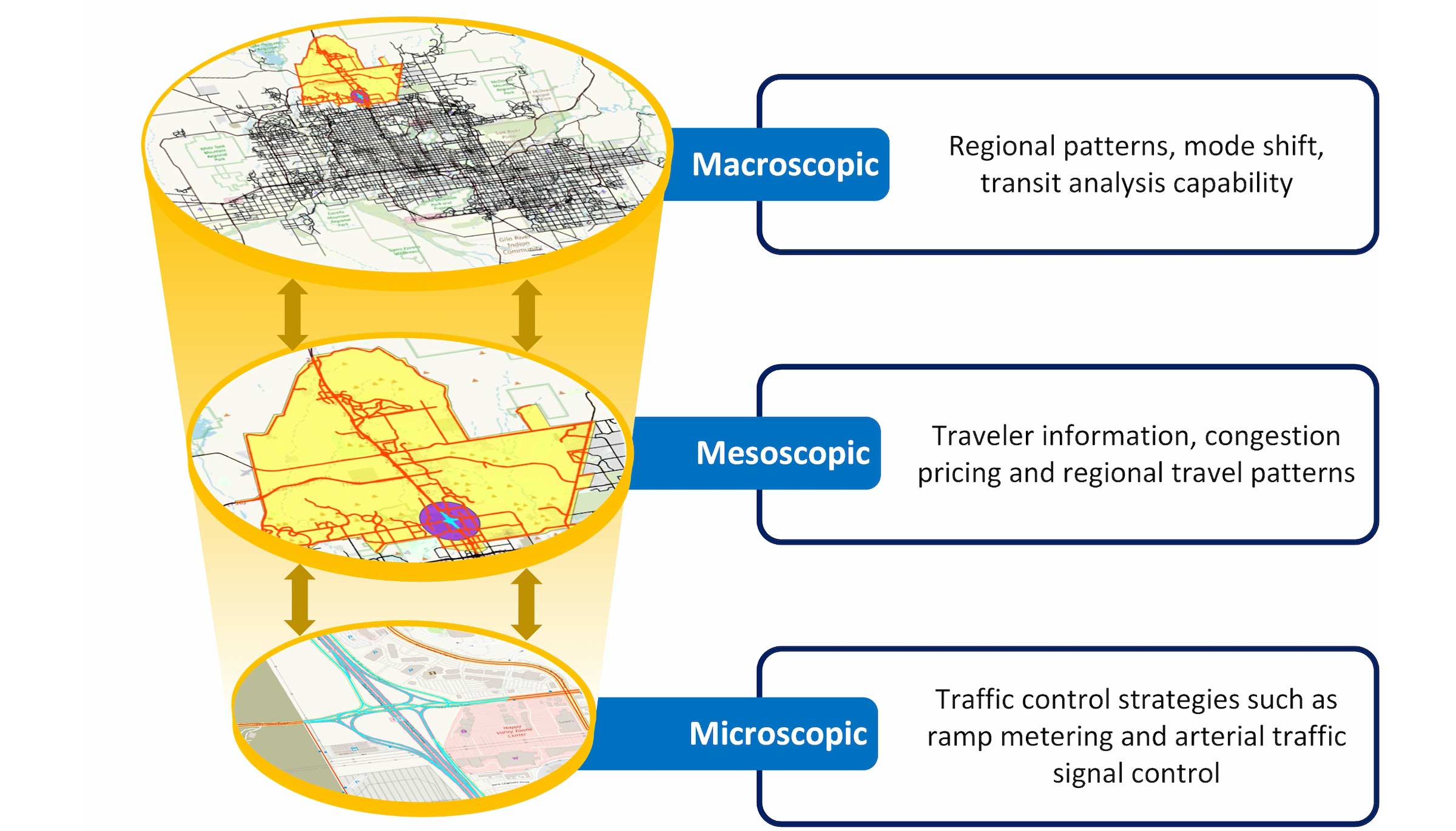
Zhou’s universal mapping system allows researchers to model transportation systems at multiple scales. Graphic courtesy Xuesong Zhou
The project is called osm2gmns, and is available to professionals around the world at no cost. It is a joint effort with Jiawei “Jay” Lu, a civil, environmental and sustainable engineering doctoral student at ASU, and many others.
In addition to creating a standard map for network analysis, Zhou’s work allows researchers to model at a more in-depth level than before with multi-level resolution. Using ASU research computing facilities, Zhou’s team also created an entire U.S. driving network from OpenStreetMap with 20 million nodes, and the related Python package has been installed more than 40,000 times.
“Traditionally, in city planning, you would look at an intersection as one point of interaction, but when you add in a pedestrian crosswalk, you now have to factor in left and right turn lanes creating more points of interaction,” Zhou says. “The same goes for adding in bike lanes; you now have to go lane by lane and determine the points of interaction among transportation system users.”
Zhou says that having an integrated transportation model at different scales including macro-, meso- and micro-levels, allows for a better cost-benefit analysis of adding various elements such as major intersections, bike lanes and even stop lights where they can be used most effectively.
“Dr. Zhou is one of the most methodologically gifted transportation scholars in the profession,” says Ram Pendyala, a fellow transportation researcher and director of the School of Sustainable Engineering and the Built Environment. “What sets him apart is that he works tirelessly to translate research into practice through the development of sophisticated software tools and systems that can be readily adopted and used in real-world planning applications.”
Supply and demand
The city of Tempe, Arizona, is characterized by multi-lane streets, bike lanes, light rail tracks and a newly added streetcar line. Due to commuting patterns, those modes of transportation can become congested as individuals start and end their work day. Determining ways to overcome this congestion and make transportation networks operate in the most time- and cost-efficient ways is a key area of Zhou’s research.
For decades, researchers have used a more traditional supply and demand curve to model the usage of transportation options. This methodology focused on one point in time and couldn’t account as accurately for multiple modes of transportation operating simultaneously in the same time.
Zhou’s most recent study, “A meso-to-macro cross-resolution performance approach for connecting polynomial arrival queue model to volume-delay function with inflow demand-to-capacity ratio,” has introduced new mathematical equations that rectify this oversight.
Associate Professor Xuesong Zhou.
“We are finally able to go from the macroscale to the microscale much more easily without using simulation,” Zhou says. “Why is this so important? Because it will save us time from doing heavy duty computational work and still provide us a precise evaluation of alternatives.
“We are still using simulations to mimic vehicle-by-vehicle congestion, but we can use this analytical tool to calculate delay time dependencies hour by hour.”
Zhou’s work in transportation modeling has sparked the interest of researchers across the world, and many are looking to Zhou for insight into their own work and the sphere of transportation research as a whole.
Zhou was recently elected to the national executive board of the Zephyr Foundation for Advancing Travel Analysis. The nonprofit organization has sought to create open-source tools, similar to those that Zhou has developed, and formulate industry standards so that transportation modeling research can be replicated easily across contexts.
“His work in modeling and optimizing dynamics of transportation networks, traffic flow and emerging mobility services is groundbreaking, and being adopted by cities and transportation planning agencies around the world,” Pendyala says.
“In the School of Sustainable Engineering and the Built Environment, we are constantly striving to advance use-inspired research that will render our future infrastructure systems more sustainable, efficient, resilient and equitable. Dr. Zhou’s work in the transportation systems analysis space contributes immensely to advancing our mission, and he is educating and training a next-generation transportation workforce fully equipped to harness the power of technology and leverage the capabilities of artificial intelligence, deep learning and operations research to achieve these goals.”
More Science and technology

Innovation Showcase opens doors for students
This May, students, faculty members, industry professionals and community supporters gathered on Arizona State University’s…

Nearly 50 years of AI at ASU
Story by Ellen ShengWhen Professor Subbarao Kambhampati started working in the field of artificial intelligence in 1983, his…

Lunar launch
When Artemis II launched in April, sending astronauts around the moon for the first time in more than 50 years, the mission broke…