Amongst the high mountain meadows and grasslands of the Ethiopian plateau stands one primate, the gelada — the “bleeding heart” monkey, named for its brilliant red chest. Geladas are the last of their kind, having outlived their extinct relatives by adopting an unusual lifestyle. Unlike their forest- and savannah-dwelling monkey cousins, geladas have carved out a high-altitude niche all to themselves, contently munching on highland grasses for sustenance.
Along with their adept mountaineering skills, geladas can typically be found in herds, clinging to cliffs in the morning, and resting on their cushion-like rumps that are ideally suited for sitting and grazing all day.
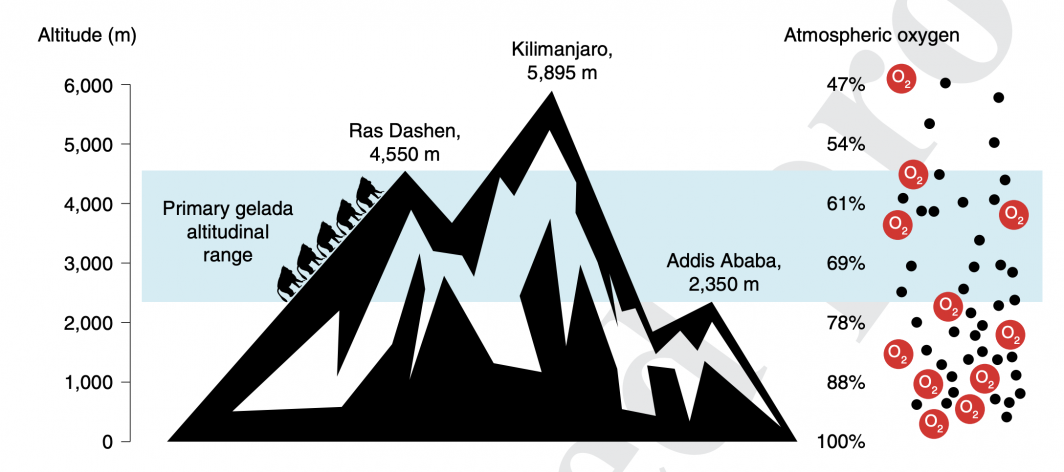
What makes them uniquely adapted to thriving in the thin airs of the plateau, 6,000–14,000 feet above sea level from their baboon cousins? And could these features have implications for human adaptation?
“Life at high altitude is very difficult. The air is colder and contains less oxygen,” said Noah Snyder-Mackler, assistant professor in Arizona State University's School of Life Sciences.
“Our team has studied geladas living in such extreme environments for over a decade, so we have a firsthand understanding of how challenging it can be to live at such heights over extended periods of time. Yet geladas have survived for much longer, making us wonder how exactly they have shifted their biology to adapt to their challenging environments.”
To crack open the molecular clues behind high-altitude adaptation, Snyder-Mackler and postdoctoral researcher Kenneth Chiou led a team to first assemble and sequence the gelada genome.
“We were interested in studying gelada high-altitude adaptations because, unlike any human population, geladas have continuously lived at high altitude for hundreds of thousands of years, giving us a window into how primates cope with extreme environments over 'deep time.'
"As geladas are closely related to us and share much of our biology, our hope is that learning more about geladas may lend insights into treatments for diseases and disorders associated with high altitude, including mountain sickness and dangerous swelling of the brain and lungs,” said Chiou, who combed the continent with colleagues to coordinate access to wild geladas and helped lead the study.
They put together the first gelada reference genome, assembled from a single wild adult female gelada from the Simien Mountains in Ethiopia, and combined it with a detailed study of data collected from wild geladas to identify any adaptations to their high-altitude environment.
The cold, thin air of the gelada habitat is a unique environmental niche, with much lower oxygen available than at sea level. Illustration courtesy Kenny Chiou
Into thin air
Within the gelada genome, the most obvious place to start was examining hemoglobins — proteins which combine in a group of four, like a magic four-leaf clover, as the key oxygen-carrying molecule found in blood.
“Many animals that have adapted to high altitude have evolved hemoglobin that can more efficiently bind oxygen, which can increase the efficiency of oxygen delivery in the bloodstream when faced with low-oxygen environments,” Snyder-Mackler said.
Sure enough, they first identified gelada-specific amino acid substitutions in the alpha-chain subunit of adult hemoglobin. They thought they had found their molecular signature of high-altitude adaptation. They found two amino acid substitutions in hemoglobin alpha, at sites 12 and 23, that are unique to geladas relative to other primates. Most importantly, these substitutions, along with all coding sequences for this protein, were found in all 70 geladas that were sequenced in the study.
But, when they tested these changes in the lab, they were in for a surprise. Remarkably, they found no differences in P50 (a measurement of the partial pressure at which hemoglobin is 50% saturated) of gelada hemoglobin compared to either humans or baboons.
“When we found unique changes in the molecular sequence of gelada hemoglobin, we thought it was a slam dunk that that these molecular changes would be the key to their ability to thrive at high altitude,” Snyder-Mackler said. “But our experiments showed that this is not the case, and provide a good reminder of the importance of experimental validation of compelling hypotheses.”
The molecular changes of the amino acid substitutions found in gelada hemoglobin simply did not appear to be associated with increased hemoglobin, or oxygen affinity. So Snyder-Mackler and Chiou turned to another potential adaptive strategy. When humans ascend to high elevation, our bodies sense the drop in oxygen and typically begin producing more oxygen-carrying hemoglobin and red blood cells in response. If geladas similarly had greater hemoglobin levels in blood, the thinking goes, perhaps they could improve oxygen transport in blood without requiring advantages in oxygen-binding of hemoglobin.
Instead, they found that hemoglobin concentrations in gelada blood were no higher than those of humans, baboons or even geladas living in zoos at sea level.
“The absence of an elevated hemoglobin concentration in wild geladas living at high altitude suggests that they can still provide enough oxygen to tissues in spite of the reduced availability of oxygen,” Chiou said. “There are a lot of other ways geladas could be physiologically compensating for low oxygen, and these could entail many kinds of changes to respiratory or circulatory traits that affect oxygen transport.”
So the team was forced to look elsewhere within the genome for high-altitude adaptations.
Next, they looked at a physiological measurement on whether the adaptation could be the result of geladas having evolved a greater lung capacity. Sure enough, from their analysis, they found high-altitude geladas exhibit significantly expanded chest circumferences, potentially allowing for greater lung surface area for increased oxygen diffusion.
“Our results show that geladas have significantly expanded relative chest circumferences compared to baboons, which parallels the larger chest dimensions exhibited by native Andean highlanders,” Chiou said.
“This finding is consistent with the possibility of expanded lung volumes, but we want to be careful, because we did not directly measure lungs in this study. We also do not yet know whether these differences are developmental responses to low-oxygen environments or due to a genetically controlled component of the adaptive toolkit in geladas.”
Ultimately, a comparison to chest dimensions in captive geladas born and reared at low altitude, which are currently unavailable, would help distinguish between these two possibilities.
ASU School of LIfe Sciences Assistant Professor Noah Snyder-Mackler in the field studying a gelada herd. Photo courtesy Kenny Chiou
To higher ground
With the hemoglobin protein picture out of the question and the lung capacity issue remaining unsolved, next, they explored any further evidence from genetic differences. They casted a wide net, across over 20,000 protein-coding genes in the gelada genome. Overall, they identified 103 genes showing signatures of positive selection in geladas that might have evolved in response to the high-altitude environment.
These included four genes involved in a hypoxia pathway (ITGA2, NOTCH4, FERMT1 and MLPH). They also identified several that have been identified as candidate genes in human hypoxia-adapted populations, including FRAS1 (which is involved in renal agenesis and exhibits adaptation signatures in Tibetans and Ethiopians). Another gene was HMBS, which is involved in heme biosynthesis in Nepalese Sherpas, and finally TNRC18, a largely unknown gene that is linked to a selection in Bajau deep-sea divers.
“While we found a lot of overlap between pathways under selection in geladas and human populations living at high altitudes, aside from notable examples listed above, few genes identified by our analysis were shared with candidate genes reported by studies of high-altitude human populations or other high-altitude primates,” Chiou said.
“This suggests that gelada adaptations to similar challenges at high altitude may largely involve adaptations in different suites of genes. In other words: Natural selection may have found a different molecular solution to the same problem.”
With the groundwork completed, they will have to continue their gene studies to further refine their understanding.
Conservation implications
Any time a new genome for an animal is completed, it paints a brand new picture of how the population came to be, and provides insights into their genetic diversity.
To better understand the demographic history of geladas, including historical population sizes and population divergence, the team also sequenced the whole genomes of 70 captive and wild geladas from multiple parts of Ethiopia (three wild central geladas; 50 wild northern geladas; and 17 captive geladas of central origin), as well as 20 hamadryas baboons from Filoha, Ethiopia.
The geladas in their sample set fell into two distinct populations that are considered subspecies: the northern population, which encompasses all wild individuals from the Simien Mountains, and the central population, which encompasses wild individuals from Guassa Community Conservation Area, as well as most individuals from zoos.
Postdoctoral researcher Kenneth Chiou led the analysis of the gelada genome to look for clues to how it adapted to high-altitude environments. Photo courtesy of Kenneth Chiou
“We found no evidence of interbreeding between the two wild gelada populations, and the genetic data suggest that these two populations have been separated from each other for around 500,000 years,” Chiou said.
They also identified a curious and very large difference in the gelada chromosomes, or karyotype, that appears to be at extremely high frequency and possibly fixed in the northern population of geladas — a chromosomal fission. They think that that the chromosomal split arose in northern geladas following this population divergence almost a half-million years ago.
The research team thinks this will be a critical piece of data for conservation efforts going forward, and may scramble the current classification of gelada species.
Given that chromosomal rearrangements tend to be associated with infertility — like mules, which are the hybrid offspring of a horse and a donkey — our findings suggest that geladas may encompass at least two distinct biological species,” Chiou said.
Chiou says this finding is important for a couple of reasons.
“First, a taxonomic revision would roughly halve the populations of each gelada species and, consequently, add urgency to conservation efforts to preserve this previously unappreciated diversity.
“Second, the split chromosome we discovered in northern geladas appears to be an extraordinarily recent example of a stable chromosomal variant in a long-lived primate. It therefore provides a unique opportunity to study chromosome evolution and the role of chromosomal rearrangements in speciation in a primate closely related to humans,” Chiou said.
Next steps
With the results from the study, the team hopes to continue to refine their understanding of gelada high-altitude adaptation. They now have many new and fruitful research avenues to explore.
“From this study, we identified a suite of gelada-specific traits that may confer adaptation to their high-altitude environment, including evidence for increased lung capacity and positive selection in a number of hypoxia-related genes and regulatory regions,” Snyder-Mackler said.
“With this in mind, our genome assembly and gelada-specific genetic changes provide multiple avenues for future research on the function of the protein-coding and regulatory changes unique to geladas. This research builds on our current understanding of the mechanisms of adaptation to extreme environments and provides an avenue for research that may have a transformative impact on the study and treatment of hypoxia-related conditions.”
The work was made possible by a large international effort from more than 30 institutions, and with the generous permissions and support from the African Wildlife Foundation, Ethiopian Wildlife Conservation Authority, the National Science Foundation, National Institutes of Health, the San Diego Zoo, University of Washington Royalty Research Fund and the German Research Foundation. Snyder-Mackler and Chiou are particularly thankful for the research staff who made the research possible (E. Jejaw, A. Fenta, S. Girmay, D. Bewket and A. Adwana), logistical support staff (T. W. Aregay and S. Asrat) and assistants and students of the Simien Mountains Gelada Research Project — especially J. Jarvey and M. Gomery — as well as the EWCA for permission and support for working in the Simien Mountains National Park.
To learn more, visit the following websites:
Simien Mountains Gelada Research Project
Guassa Gelada Research Project
San Diego Zoo Frozen Zoo
The study appears in the journal Nature Ecology and Evolution (DOI: https://doi.org/10.1038/s41559-022-01703-4).
Top photo: Among the high mountain meadows and grasslands of the Ethiopian plateau stands one primate, the gelada — the “bleeding heart” monkey, named for its brilliant red chest. Geladas have carved out a high-altitude niche all to themselves. Photo courtesy India A. Schneider-Crease/ASU
More Science and technology
Space no longer the final frontier
As the Aerospace and Defense Summit got underway on May 15 in Los Angeles, the purpose of the summit was made abundantly clear.“Space is no longer the frontier,” said Anna Magzanyan, president of LA…

ASU researchers zero in on how sudden stress drains bacterial energy
When bacteria are suddenly hit with a salty or sugary shock, they do not just shrivel slightly and carry on. According to new research from Arizona State University, they also suffer a rapid drop in…

ASU joins Applied Materials' EPIC Center as inaugural university research partner
When the United States made the strategic decision in 2020 to reinvest in domestic semiconductor manufacturing, it confronted an uncomfortable paradox baked into capitalism itself: How do you create…