Walking with coffee is something most of us do every day without considering the balancing act it requires. In fact, there’s a lot of physics preventing the coffee from spilling over.
The coffee, a thermally agitated fluid contained in a cup, has internal degrees of freedom that interact with the cup, which, in turn, interacts with the human carrier.
“While humans possess a natural, or gifted, ability to interact with complex objects, our understanding of those interactions — especially at a quantitative level, is next to zero,” said Ying-Cheng Lai, an Arizona State University electrical engineering professor. “We have no conscious ability to analyze the influences of external factors, like noise or climate, on our interactions.”
Yet, understanding these external factors is a fundamental issue in applied fields such as soft robotics.
“For example, in design of smart prosthetics, it is becoming increasingly important to build in natural modes of flexibility that mimic the natural motion of human limbs,” said Brent Wallace, a former undergraduate student of Lai’s and now a doctoral student in ASU’s Ira A. Fulton Schools of Engineering. “Such improvements make the prosthetic feel more comfortable and natural to the user.”
According to Lai, it is conceivable that, in the not-too-distant future, robots will be deployed in various applications of complex object handling or control, which require the kind of coordination and movement control that humans do quite well.
If a robot is designed to walk with a relatively short stride length, then relatively large variations in the frequency of walking are allowed. However, if a longer stride is desired, then the walking frequency should be selected carefully.
A new paper published in Physical Review Applied, “Synchronous Transition in Complex Object Control,” originated with Wallace as part of his senior design project in electrical engineering, supervised by Lai. Wallace has received an NSF Graduate Fellowship and now is a doctoral student in ASU’s School of Electrical, Computer and Energy Engineering.
The ASU team’s research expands on a groundbreaking virtual experimental study recently conducted by researchers at Northeastern University, using the coffee-cup-holding paradigm and adding a rolling ball, to examine how humans manipulate a complex object. Participants deliberately rotated the cup in a rhythmic manner with the ability to vary force and frequency to ensure the ball stayed contained.
The Northeastern study showed that the participants tend to select either a low-frequency or a high-frequency strategy — rhythmic motion of the cup — to handle a complex object.
A remarkable finding was that when a low-frequency strategy was used, the oscillations exhibit in-phase synchronization, but antiphase synchronization arises when a high-frequency strategy was employed.
“Since both the low- and high-frequencies are effective, it’s conceivable that some participants in the virtual experiment switched strategies,” Wallace said. “This raises questions.
“How does a transition occur from in-phase synchronization associated with a low-frequency strategy to antiphase synchronization associated with a high-frequency strategy, or vice versa?” Wallace asked. “In the parameter space, is the boundary between the in-phase and antiphase synchronization regimes sharp, gradual or sophisticated?”
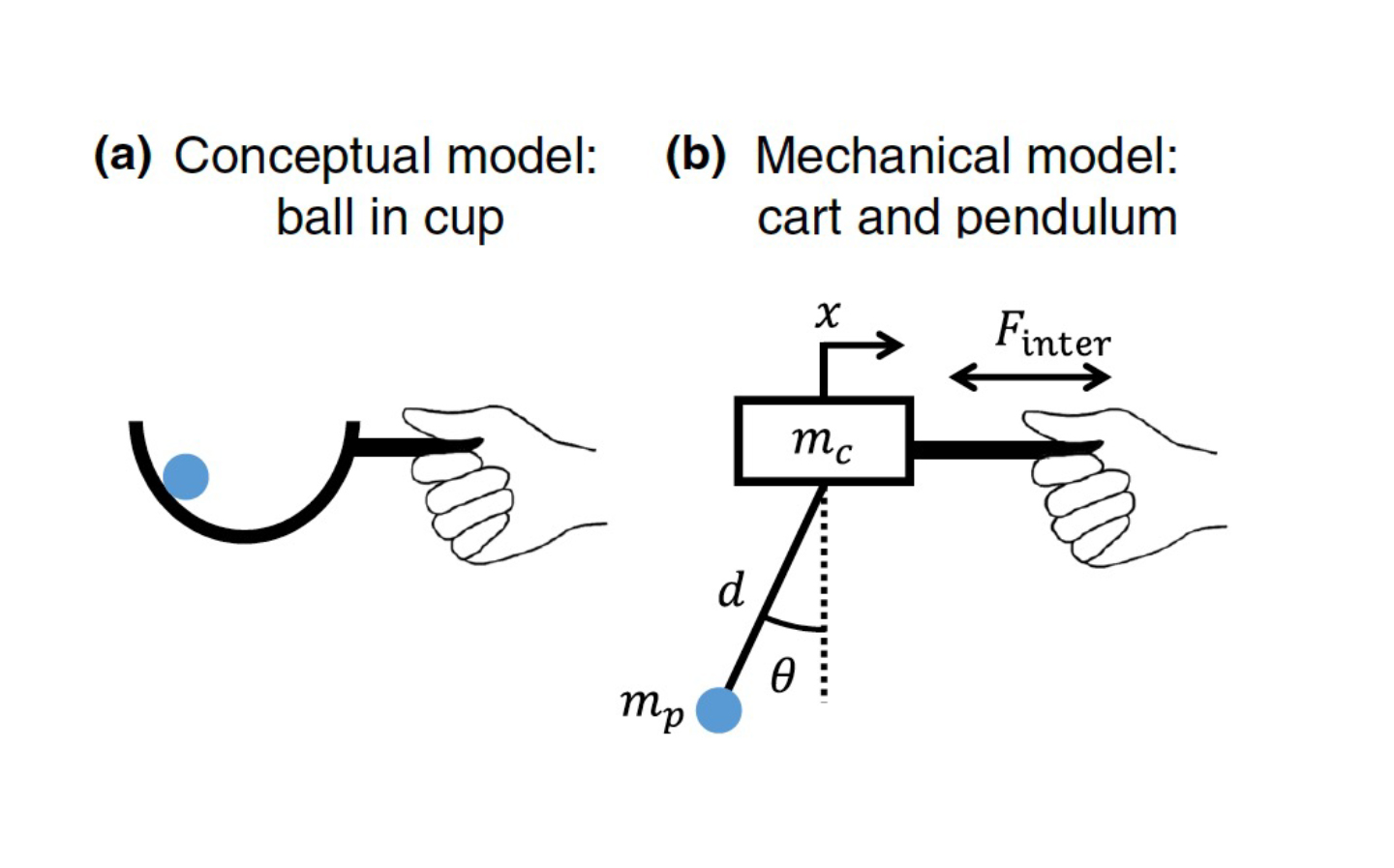
The ASU team’s research, prompted by Wallace’s curiosity, studied the transition between the in-phase and antiphase synchronization using a nonlinear dynamical model of a pendulum attached to a moving cart subject to external periodic forcing.
A schematic illustration of the cart-pendulum system to simulate a human’s handling of a complex object, such as a cup of hot coffee: (a) a conceptual model of a ball rolling inside a circular cup and (b) a nonlinear mechanical model of a pendulum attached to a moving cart, which is mathematically described by a set of differential equations.
The researchers found that, in the weakly forcing regime, as the external driving frequency is varied, the transition is abrupt and occurs at the frequency of resonance, which can be fully understood using the linear systems control theory.
Beyond this regime, a transitional region emerges in between the in-phase and antiphase synchronization, where the motions of the cart and the pendulum are not synchronized. It was also found that there is bistability in and near the transitional region on the low-frequency side.
Overall, the results indicate that humans are able to switch abruptly and efficiently from one synchronous attractor to another, a mechanism that can be exploited for designing smart robots to adaptively handle complex objects in a changing environment.
“It is possible that humans are able to use both in-phase and antiphase strategies skillfully and to switch from one strategy to another smoothly, perhaps without even realizing it. The findings from this study can be used to implement these human skills into soft robots with applications in other fields, such as rehabilitation and brain-machine interface,” Lai said.
Additionally, tasks as trivial as running wires in a car body on an assembly line — which humans carry out with ease — still elude the most advanced machines.
“A systematic quantitative understanding of how humans interact dynamically with their environment will forever change how we engineer our world, and may revolutionize the design of smart prosthetics and usher in new age of manufacturing and automation,” Wallace said. “By mimicking the dynamically favorable behaviors adopted by humans in handling complex objects, we will be able to automate processes previously thought to be impossible.”
Additional authors include Ling-Wei Kong and Armando Rodriguez, also from ASU.
DOI: 10.1103/PhysRevApplied.16.034012
Top image by iStock
More Science and technology
Space no longer the final frontier
As the Aerospace and Defense Summit got underway on May 15 in Los Angeles, the purpose of the summit was made abundantly clear.“Space is no longer the frontier,” said Anna Magzanyan, president of LA…

ASU researchers zero in on how sudden stress drains bacterial energy
When bacteria are suddenly hit with a salty or sugary shock, they do not just shrivel slightly and carry on. According to new research from Arizona State University, they also suffer a rapid drop in…

ASU joins Applied Materials' EPIC Center as inaugural university research partner
When the United States made the strategic decision in 2020 to reinvest in domestic semiconductor manufacturing, it confronted an uncomfortable paradox baked into capitalism itself: How do you create…