Using LED lights, NeoLight aims to help tens of thousands of newborns every year
Each year, around 60% of newborns will come into the world with jaundice, a condition that manifests as a yellowing of the baby’s skin. Infant jaundice is easily treatable and is often treated with phototherapy, which involves exposing the newborn to blue light for a few days after birth. But in parts of the developing world that lack access to electricity or medical supplies, tens of thousands of infants die or develop brain damage each year from untreated jaundice.
Sivakumar Palaniswamy, a 2016 Arizona State University graduate with an MS in bioengineering, saw this tragedy firsthand while working in a hospital in India, where several newborns shared a single light. In the summer of 2014, Palaniswamy partnered with three then-fellow ASU students to found NeoLight, a medical startup aimed at eradicating deaths from infant jaundice with a new approach to phototherapy.
“Siva saw a problem and it struck a chord in his heart,” said NeoLight CEO Vivek Kopparthi, ’14 MS in management. “When he returned to ASU, we started discussing this and applying the biomedical concepts we learned in school to engineer an efficient solution.”
NeoLight got an early boost with a $25,000 grant from ASU’s Edson Student Entrepreneur Initiative. The team parlayed the grant into a further $600,000 seed investment from angel investors in Phoenix that they used to navigate the complex regulatory approval process for medical devices; in 2017, the device was approved by the FDA. To date, the company has raised around $7.5 million from investors, including NFL quarterback Ben Roethlisberger, and has developed a partnership with Phoenix-based HonorHealth to further study the device.
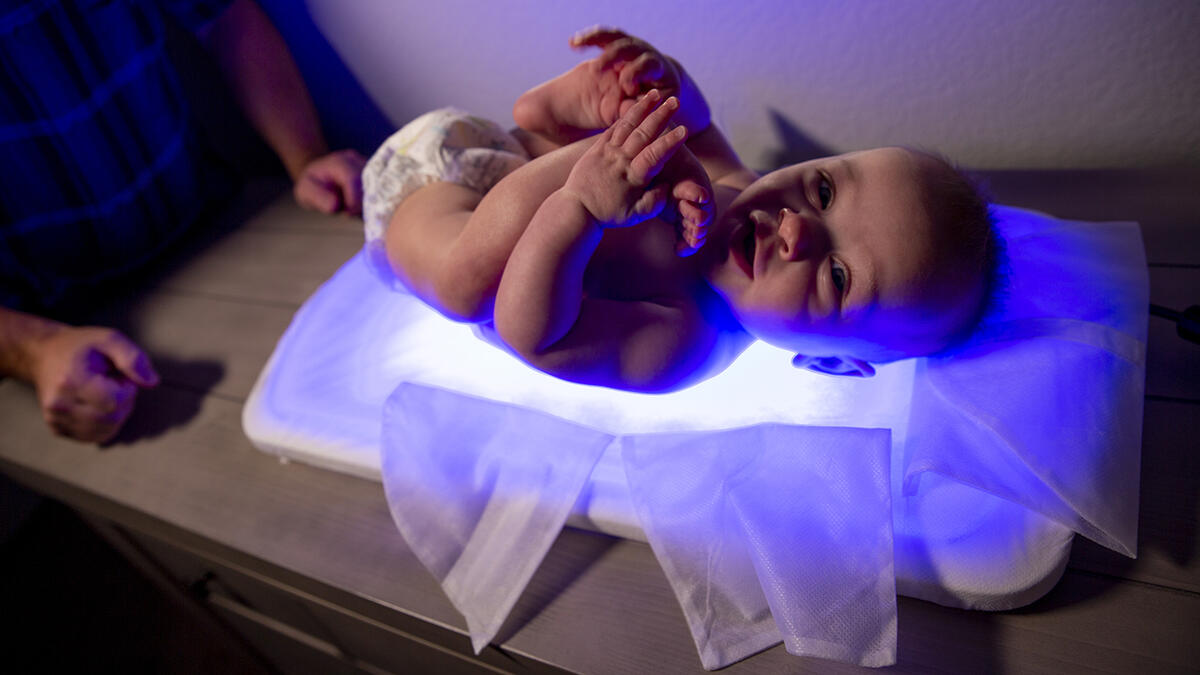
Known as Skylife, the device uses LED-based phototherapy to treat infant jaundice in both hospitals and homes. LEDs are highly efficient, using 98% less electricity than a standard incubator, so they can be used in places without reliable electricity, and they don’t have many of the side effects of other phototherapy devices like excess heat that can dry a baby’s skin. Furthermore, the device has been shown to send jaundiced babies home from the hospital 40% faster than competitors’ products.
The company is also developing a diagnostic device that detects and monitors jaundice on the spot in newborns without the need for a lab test. With these devices, the NeoLight team hopes it will soon make untreated infant jaundice a thing of the past.
Written by Daniel Oberhaus. He is a graduate of Barrett, The Honors College who earned a Bachelor of Arts in creative writing and philosophy in 2015, a staff writer at Wired magazine and the author of “Extraterrestrial Languages” (MIT Press). This story originally appeared in the spring 2020 issue of ASU Thrive magazine.
Top photo: The FDA-approved Skylife, shown here under a baby, treats newborns for jaundice in homes or hospitals. The founders won a $25,000 grant from ASU’s Edson Student Entrepreneur Initiative and parlayed that into an additional $600,000 seed investment. Photo by Erika Gronek/ASU
More Science and technology
Extreme HGTV: Students to learn how to design habitats for living, working in space
Architecture students at Arizona State University already learn how to design spaces for many kinds of environments, and now they can tackle one of the biggest habitat challenges — space architecture…

Human brains teach AI new skills
Artificial intelligence, or AI, is rapidly advancing, but it hasn’t yet outpaced human intelligence. Our brains’ capacity for adaptability and imagination has allowed us to overcome challenges and…

Doctoral students cruise into roles as computer engineering innovators
Raha Moraffah is grateful for her experiences as a doctoral student in the School of Computing and Augmented Intelligence, part of the Ira A. Fulton Schools of Engineering at Arizona State University…